- Home
- About
- Corporate
- Products
- Shoulder Sports Medicine
- Knee Sports Medicine
- ACL-PCL Implants
- ACL-PCL Instruments
- Onloc
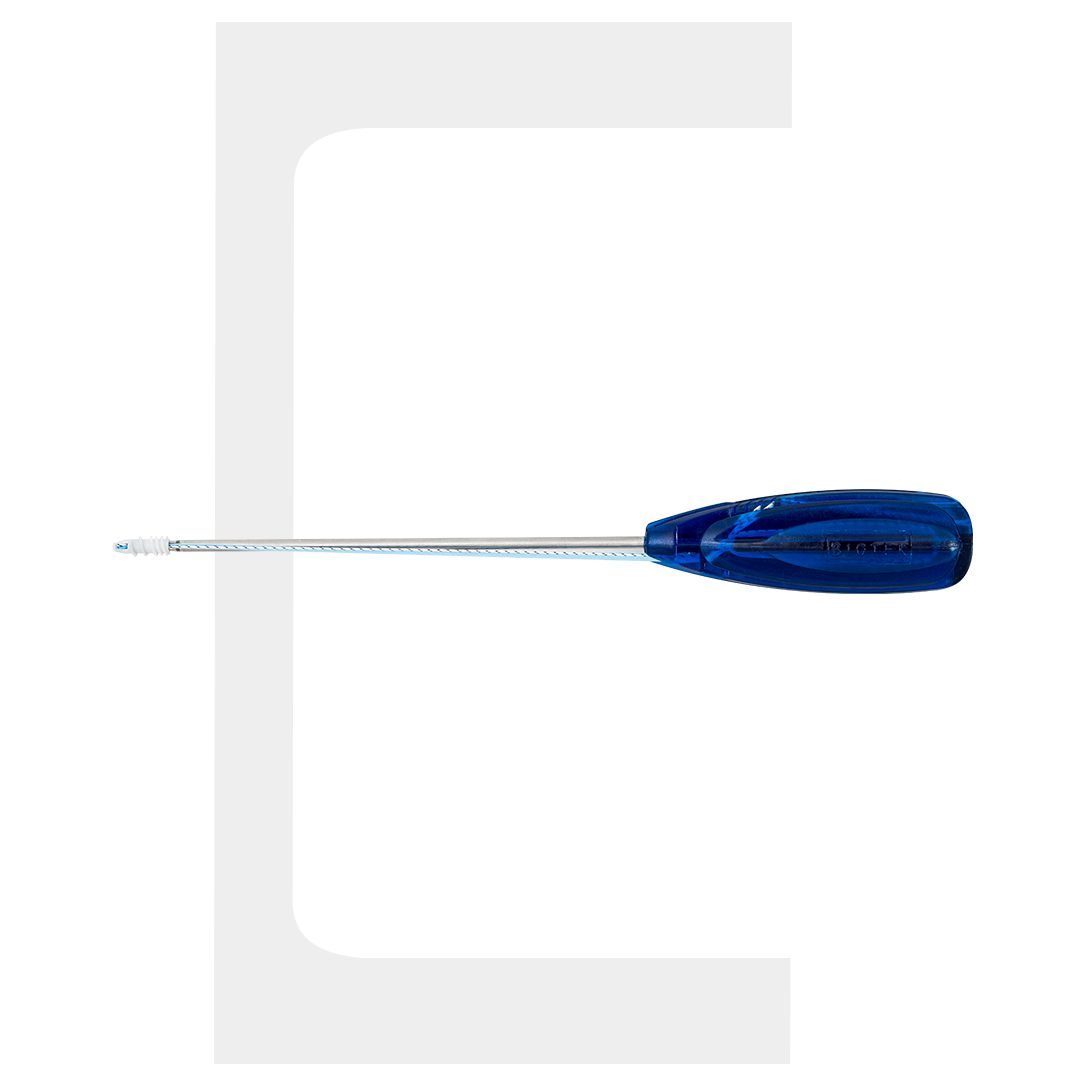
- Drill Bit Passing Pin
- AI Reamer
- Flowertip
- Cannudrill
- Flexi
- Shuttle
- Depth Guage
- Sureshot
- Locus
- Bone Tunnel Plug
- Tunnel Dilator
- Arthroscopic Probe
- Microfracture Awls
- Graft Sizing Block
- Rasps
- Tunnel Notcher
- Graft Prep Station
- Tendon Stripper
- Notchplasty
- PCL Elevator
- PCL Rasps
- PCL Curette
- Graphic Case
- Meniscus Repair Implants
- Meniscus Repair Instruments
- Elbow Sports Medicine
- Foot & Ankle
- Osteotomy
- Hand & Wrist
- Extremities
- Hip Sports Medicine
- News & Events
- Contact